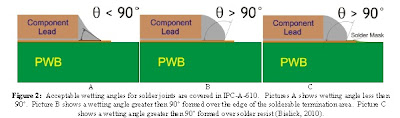
Experiment Process
One of the major goals of this study was to control as much as possible the previous factors with the purpose of observing any difference in wetting behavior caused by the reflow technique. The samples used as the solderable metal surfaces were 10 Cu Blocks. Each Cu Block had the dimensions: 38 mm X 38 mm X 3.2 mm. Figure 4 shows an example of the Cu Block Sample.
Five of the Cu Blocks went through Vapor Phase Reflow and five of the blocks went throug. Convection Reflow. A separate Cu Block had three thermocouples (TC’S) attached to it and was used to set up the recipes of the thermal reflow processes.
The recipes developed were optimized for Pb Free SAC soldering parameters. All three TC’s were attached to the topside surface and were adhered from one corner to the opposite corner. The Vapor Phase Reflow machine was manufactured by R&D Technical Services. The Vapor Phase machine was a batch unit that had a fully enclosed topside/bottomside convection heat pre-heat zone, and a reflow zone containing the vapor well. Both in the pre-heat and reflow zone Nitrogen (Inert) gas was supplied. The following was the recipe used:
- Boiling Fluid – 240 perfluorinated heat transfer fluid
- Pre-Heat - Oven at 290°C for 175 Seconds
- Reflow Dwell – 115 Seconds
- Vapor Flash Off Dwell – 25 Seconds
- Cool Dwell – 400 Seconds
Figure 5 shows the corresponding thermal profile on the Cu Block.
The Convection Reflow machine used was an inline unit from Heller Industries. The Convection
Reflow oven had 12 Topside/Bottomside Zones and had Nitrogen (Inert) gas atmosphere. The Nitrogen source was same for both the Vapor Phase and Convection Reflow Machines. The following was the recipe used:

Figure 6 shows the corresponding thermal profile on the Cu Block. Table 1 compares thermal profile measurements between the two reflow processes.


As shown in Table 1, the Convection Reflow profile peaked about 5 degrees hotter and stayed about 15 seconds longer above 217°C then the Vapor Phase Reflow profile. Note for the heating ramps and cooling ramps, both reflow processes were equivalent. For this experiment, the greater amount of heat and time above the liquidous was assumed to have minimal effect in reflowing the solder and solder solidification.


For sample preparation, each Cu Block was cleaned using a Scotch Brite (Trademark of 3M Corporation) Pad, followed by Xylene and Isopropanol wipe, and allowed to dry. Figure 4 shows the Cu Block metal surface after cleaning. A SAC 305 Pb free solder paste was used for the solder deposition. Here are the data specifications for the solder paste (Indium, 2008):
- 96.5Sn/3.0Ag/0.5Cu (SAC305)
- Type 3 Mesh
- No clean Flux Chemistry – Flux Type R0L0
- Halide-Free
- Typical Solder Paste Viscosity (Malcom 10 RPM) – 1700 poise
- Typical Tackiness – 35 g
A manual mini-stencil was used to deposit the solder paste on each Cu Block. The 0.152 mm thick stencil had 0.508 mm diameter apertures on 1.27 mm pitch for a total of 624 apertures. Figure 7 shows an example of the solder paste deposition on a Cu Block Sample. After solder dispensing the Cu Samples were immediately run through the corresponding reflow process.

Optical Results of Solder Bumps Post Reflow
Figures 8 and 9 show the typical solder bump results on each Cu Block sample post reflow. Each figure includes an (a) and (b) picture, where the (a) picture was from the Vapor Phase Reflow, and the (b) picture was from the Convection Reflow.
From an optical / qualitative perspective, the Vapor Phase deposits appear to have larger area coverage than the corresponding Convection Reflow samples. Also with the Vapor Phase Reflow Solder deposits a larger halo of silver/gray material formed around the bump, which is shown in Figure 9. This halo was confirmed by EDX as Sn and is shown in Figure 10. One hypothesis is that at some point during the vapor phase reflow process the solder liquid had spread further in area but during solidification the solder perimeter pulled back leaving Sn metallization on the Cu Surface. Another hypothesis is that Vapor Phase Reflow caused a greater slumping/spreading of the solder paste, then the coalescence of solder paste occurred followed by a pull back of the molten solder boundary to a state of stable equilibrium (Bielick, 2010). This Sn haloing was not observed as much on the Convection Reflow solder deposits. Also observed with the Vapor Phase samples was a greater amount of transparent residues and the Cu Blocks appeared to be more tarnished from Vapor Phase than Convection Reflow. One last visual observation about all the samples was that there was variability in the amount of solder, specifically that the solder bumps were smaller near the corner and edges than the solder bumps in the middle of the array.